We enter 2026 as a new reality is forming. It will not be “business as usual”. Geopolitical forces in the largest market for pharmaceuticals, the United States, are likely to mean substantial changes. In this article we cover those changes, the incretin juggernaut and finally the march of technical progress with “minitablet” dispensers, large volume injectors, and dual chamber devices.
Changes in the US market
The United States accounts for around 45% of global pharmaceutical sales by value[1], and the majority of global pharmaceutical profits. Donald Trump has announced a policy of “Most Favoured Nation” pricing for pharmaceuticals with aim of making drug pricing for people living in the United States no more than people living in comparable G7 countries.
In addition, there is an “America first” policy to onshore pharmaceutical manufacturing. Major pharmaceutical companies have responded by announcing significant investments in US manufacturing sites[2],[3]
While pharmaceuticals were exempt from the tariffs announced on 2nd April 2025, the Bureau of Industry and Security, initiated a Section 232 national-security investigation into pharmaceutical imports[4]. The White House has subsequently stated that tariffs on pharmaceuticals entering the United States will be introduce[5]. The United Kingdom has signed a trade deal to avoid tariffs on its pharmaceutical exports to the United States in return for increased spending on drugs[6].
Finally, the FDA vaccine committee has been replaced by new appointees with known scepticism of some vaccine technologies such as mRNA vaccines.
Altogether, if these changes take effect before the next US administration enters office in 2029, they represent substantial changes to the way medicines are reimbursed, taxed, regulated, and where they are made. The same effects will be felt through the supply chain, affecting drug delivery device manufacturers, packaging suppliers, fill-finish partners and others. In this context our company Sanner Group has expanded operations into the United States to provide device development, manufacturing, and Human Factors engineering on American soil in addition to our capabilities in Europe and China. Companies throughout the drug delivery ecosystem need to consider multinational strategies or face significant disruption to their business and/or barriers to growth.
The incretin juggernaut
Incretin receptor agonists such as glucagon-like peptide (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) have proven to be clinically effective in managing type 2 diabetes mellitus and obesity. The demand for incretin receptor agonists has led to a rapid growth in their use with the US seeing a 700% increase over four years in number of patients without diabetes starting treatment[7]. Two phenomena have accelerated in the incretin boom. First, direct-to-consumer sales of injection devices have become “mainstream” such as being promoted by social media influencers and prominent in modern decentralized media. Celebrities such as Elon Musk, Oprah Winfrey, Sharon Osbourne, Serena Williams have spoken publicly about using incretins for weight management. This brings cultural acceptance and even advocacy [See Figure 1.]

Figure 1: Example use of incretin agonist injector by celebrity (in this case Serena Williams). Source: MEGA
Second, “compounding” pharmacies have become far more prevalent in the market[8]. Compounding pharmacies combine or alter ingredients to create a customized medication for an individual patient based on a licensed practitioner’s prescription.[9] They use FDA-approved ingredients, but the resulting formulation is not FDA-approved.[10] Therefore, compounded medicines are regulated differently from the originator and may vary in quality. An example compounded formulation is the addition of vitamin B12 to semaglutide. Compounding pharmacies have manufactured large quantities of semaglutide since May 2023 when the FDA placed Ozempic and Wegovy on their Drug Shortages List because demand outstripped the quantities that Novo Nordisk could supply to the US market.[11] Semaglutide was removed from the Drug Shortage List in February 2025[12], but the compounders have continued marketing semaglutide to the extent the market for compounded semaglutide in the US is “roughly equal” in size to Novo Nordisk’s sales of that drug in that market.[13] This is effectively generic/biosimilar competition before the originator patents expire, which changes significantly the economic viability for developing new medicines. Balancing pricing with cost pressures from both competitors and competitor generics can be a challenge with CVS Caremark reportedly removing Zepbound from its formulary in July of this year in a move to balance cost with access to these therapies[14]. Furthermore, while compounding pharmacies typically cannot supply the same drug delivery device used by the originator. This isn’t the case for counterfeit products which are also springing up on social media. UK regulators uncovered an illicit supply of counterfeit Retatrutide (the AbbVie drug undergoing clinical trials) and Tirzepatide being sold with pen-injector devices by entities on social media[15]. Despite the seizure of thousands of unlicensed pens and raw materials, sellers reportedly continue to promote these unregulated products online, driven directly by consumer demand for these products.
Looking ahead, the market may become increasingly stratified. Longer acting formulations, including monthly or even quarterly dosing, have undergone animal trials[16]. A push towards multi-agonist “Triple G” parenteral therapies such as Retatrutide may also result in therapies which require devices to administer larger weekly injections of up to 1.0 ml[17].
Equally, multiple oral dosage forms targeting the treatment of obesity, such as Lilly’s orforglipron, are in development. While also offering lower cost and greater access, the availability of oral therapies provides a new option within the class for patients who are unable or unwilling to self-administer an injectable agent. The shorter half-lives may benefit patients who need to adjust their dosage to counteract side effects.
Smart dispensers and minitablets
Many medicines have better efficacy and safety profiles (better outcomes with less side effects and adverse events) if they are personalised to the patient[18].This can be particularly true where the patient population is inherently highly diverse such as in paediatrics. Children vary greatly in their body weight, metabolism, tolerance, and anatomy. Blanket age range recommendations can be undermined by the significantly different growth rates experienced by children.
In addition, some patients find it difficult to swallow the pills and tablets used for many medicines. Sometimes compounding pharmacies are used to create alternative presentations of the drug substance (see above) but there could be a case for formulating drugs as powders or minitablets to be metered at the point of use.
Finally, some medicines need to be “titrated”, which means beginning with a low dose and gradually adjusting it upwards (or sometimes downwards) until the desired therapeutic effect is reached whilst minimising side effects. Reasons for titration include:
Safety: some drugs can cause serious side effects if started at full strength. Titration gives the body time to adapt.
Individual variability: patients respond differently to the same drug depending on age, weight, metabolism, other medications, or medical conditions.
Therapeutic window: Some drugs have a narrow range between being effective and being toxic. Titration helps find the “sweet spot.”
Monitoring response: doctors can observe how symptoms improve and adjust accordingly.
The drug delivery industry has long provided devices such as pen injectors to allow user-settable doses of injectable drugs such as insulin, GLP-1, growth hormone, and so on.
Fine-increment user-settable dosing is less well established in oral drug delivery. Several devices are in development to meet the need for variable dosing. For example, “Diffucaps + POWDOSE” is a combination system where the POWDOSE device dispenses the desired number of minitablets, and the Diffucaps polymer membrane encases the minitablets to control drug release and enhanced solubility in targeted gastrointestinal regions.[19]
OraFID by Sensidose (part of Navamedic) is a prefilled, single-use container and dispenser where the user twists a counter counterclockwise then clockwise and presses a button to dispense the desired number of minitablets.[20] Mini-tablet Dispenser by Phillips-Medisize is a cap which fits onto a standard table bottle and helps the user see the number of minitablets about to be dispensed.[21] Finally, OnDosis is an electro mechanical minitablet dispenser with the option of syncing dispense data with a smartphone app[22]. This list gives some examples and is not intended to be exhaustive.
There are also developments in 3D printing of tablets and modular Oral Solid Dose (OSD). Modular manufacturing means using standardised, flexible production units that can be scaled up or down depending on requirements. The aim is faster adaptation to different drug formulations, easier compliance with regulatory requirements, and efficient production of multiple dosage forms in one facility.
Continued growth of large volume injections
The market for large-volume autoinjectors and on-body delivery systems has expanded rapidly as injectable therapies become more complex and dose masses continue to increase. One driver for this is an industry trend toward reformulating intravenous therapies for subcutaneous delivery, particularly in oncology and immunology[23]. Well-known examples such as Keytruda, Darzalex, Rituxan, Herceptin, and Skyrizi illustrate this shift, driven by patient convenience, reduced healthcare burden, and improved access.
For biologics, particularly monoclonal antibodies, high dosage mass often translates into either highly viscous formulations, large injection volumes, or both. These physical properties place significant demands on delivery devices and driving innovation as devices increasingly need higher power (See Table 1), or longer and slower delivery times which favour on-body devices.
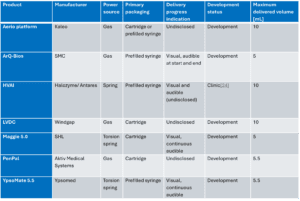
Table 1 – Overview of large volume autoinjector technologies
The increasing power requirements of large volume and high viscosity injections have led many companies to explore gas pressure as a means of driving the delivery mechanism. Gas powered actuation is not new, but it addresses several inherent limitations of traditional spring driven autoinjector designs.
One key limitation of springs is the dynamic impact associated with releasing large amounts of stored mechanical energy, which can generate force peaks and increase the risk of glass breakage. Gas based systems can deliver high power in a more controlled and progressive manner using valving or expansion chambers, avoiding the sharp force transients that occur when a spring impacts the plunger stopper. Managing high internal pressures remains challenging when working with glass primary packaging, and one approach taken by Aktiv in its PenPal device is to use the outer casing as a pressure chamber, pressurising both the inside and outside of the primary container. This reduces differential pressure across the glass wall and mitigates hoop stress.

Figure 2 – PenPal by Aktiv medical systems
Compared with spring driven mechanisms, gas powered systems can also deliver a much flatter force profile throughout the injection. This improves control of the delivery rate and is particularly important for concentrated biologics and long acting injectables (LAIs), which often exhibit non-Newtonian behaviour and therefore an apparent viscosity that varies with applied pressure.
Windgap has demonstrated that gas actuation can also enable reciprocal motion, making it well suited to applications such as automated reconstitution and multi dose delivery, where pausing, restarting or cyclic movement may be required.
From a platform perspective, gas powered systems offer a high degree of flexibility and adaptability. Delivery performance can often be tuned by adjusting the gas fill, measured gravimetrically in production, rather than requiring redesign of springs. Common propellants include gases such as argon and nitrogen, which are relatively insensitive to environmental variability and capable of delivering very high pressures, as well as subliming gases stored in a liquid phase, such as carbon dioxide and hydrofluoroalkanes (HFAs). These provide lower pressures but a consistent force profile, subject to stable ambient temperature and pressure.
Given the growing interest in large dose subcutaneous administration, this segment can be expected to develop. Tolerability of large injections remains a significant challenge, although this may be partially addressed using adjuvants such as hyaluronidase. In this context, the inclusion of a 10 ml autoinjector from Halozyme should be viewed as a strong signal of the company’s confidence in the viability of this as a delivery approach.
Dual chamber injectors
Dual-chamber technologies have traditionally been developed to support lyophilised drug products, where the drug is reconstituted with a diluent prior to injection. The dual-chamber format is well established and widely accepted because it simplifies preparation whilst maintaining stability during storage. However, dual chambers are not well suited to liquid–liquid co-delivery because they commonly rely on the compression of a significant air volume before the stopper reaches the bypass that connects the two chambers. The need for this air volume limits compatibility with larger liquid-liquid fill, and standard fill-finish processes.
Several solutions are being developed to support therapies that require the controlled delivery or mixing of two liquid components immediately prior to injection. This need arises where liquid–liquid stability is limited or unproven, where an excipient, buffer, or stabiliser must be introduced shortly before administration, or where formulations benefit from remaining physically separated until the point of use. There is a split between the development of new dual chamber primary packaging or devices that leverage standard primary packaging, achieving sequential delivery from two separate containers.
Kindeva’ s DuoDote® is a commercialised example of a dual-liquid delivery system. Used for the emergency treatment of nerve agent poisoning, the device contains atropine and pralidoxime chloride in two separate chambers and delivers them sequentially through a single needle in one activation. The product is FDA-approved and, in early 2025, Kindeva Drug Delivery’s Meridian Medical Technologies division secured a significant contract to supply DuoDote® to the U.S. Strategic National Stockpile[25]..
Windgap has developed dual-liquid autoinjector platforms based on separate reservoirs rather than a single dual-chamber syringe with a mechanical bypass. This approach enables controlled sequential or coordinated delivery of two liquids through a single needle. The architecture is particularly well suited to molecules that need to be isolated because of specific fill-finish or packaging requirements. large-volume and high-viscosity applications, and can be used to perform reconstitution.
Credence MedSystems[26] Dual Chamber Syringe System (DCS) enables multiple liquids to be delivered in sequence with a single press of the plunger rod. The system also features a proprietary passive needle retraction system and has recently been adapted into a new autoinjector concept.
Capa Valve[27] has developed a patented valve system that can be installed into standard syringes to create dual-chamber functionality without needing bespoke primary packaging. The valve technology allows two liquids (or a liquid and a diluent/powder) to be stored separately and then dispensed sequentially through a single syringe. This approach leverages established infrastructure, minimising manufacturing complexity.
BD has explored dual-liquid syringe concepts that maintain compatibility with standard prefillable syringe formats[28]. They recently published a white paper on the BD Dual-Injection Valve which similarly keeps two liquid components separate within a single primary container.
This is an interesting area of innovation which may see growth as drug-drug and drug-biologic combination therapies become more prevalent due to their enhanced effectiveness, ability to tackle complex diseases like cancer, chronic conditions and facilitate personalised medicine where the combination is adjusted to a specific sub-set of the population[29].
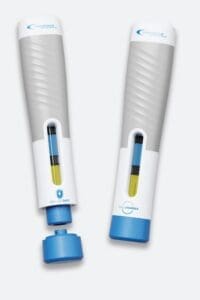
Figure 3 – Credence Medical autoinjector concept which includes Credence’s Sequential Dual Chamber System
As we enter 2026, the pharmaceutical and drug delivery landscape is entering a period of fundamental change. Geopolitical developments are reshaping how medicines are priced, regulated, manufactured and supplied, with effects that will be felt globally. For companies across the drug delivery ecosystem, these shifts demand a reassessment of their long-term strategy.
The continued rapid expansion of the incretin receptor agonist market has reshaped not only pricing strategies but also patient expectations and the nature of competition. The emergence of early generic like competition and counterfeit products adds further complexity, while longer acting injectables, multi agonist therapies and oral alternatives point to sustained innovation in this therapeutic area for many years to come.
We see device technologies evolving to meet future needs. Variable dosing systems, smart dispensers and minitablet based approaches address the growing requirement for personalisation, particularly for diverse or vulnerable patient populations. Dual chamber liquid-liquid injection further expands the available toolkit, enabling the delivery of more complex and potentially personalised therapies. At the same time, the continued growth of large volume subcutaneous delivery is driving innovation in high power autoinjectors as an alternative to on body systems, although the tolerability of such injections remains an open question.
Taken together, these trends suggest that innovation remains strong across the industry, yet there is no one size fits all solution. Instead, success is increasingly defined by adaptability to the needs of patients, pharmaceutical companies, payers, and regulatory authorities.
If you would benefit from the contract development or manufacturing capabilities of Sanner Group, please do not hesitate to get in touch with the authors.
Written by: Tom Oakley, VP Strategic Partnerships and Alex Vasiev, Head of Drug-Device Integration
Drug Delivery Trends 2026 reference guide