New injection devices are being asked to deliver larger volume injections, with higher viscosity formulations, and with increasing expectations of users on device aesthetics, simplicity, and quality. All injection devices risk finding that they fail to perform adequately when challenged with components, volumes, and viscosities at the upper end of their tolerances. The risk of failure is present when developing devices for biosimilars and generics too.
Therefore, it is becoming ever more important to predict a device’s behaviour early in the development, and to predict the effect of shelf-life ageing. However, with increased drug viscosity and volume comes more complex force requirements, and basic methods of predicting plunger forces are not sufficiently capable.
The nightmare scenario is that an injection device is designed, taken through tooling and automation, and design verification testing, and then found to fail its performance requirements after shelf-life.
Here we describe a new method and enabling software that we developed to enable engineers to translate basic syringe test data into injection device performance, and to work backwards from device performance after shelf-life to the maximum forces they can allow in a syringe test. This can be done early in development to avoid failures late in the program.
Why use Break-Loose Extrusion force data?
Break-loose and extrusion force (BLEF) tests are a common way to characterize prefilled syringes (with plungers). In a BLEF test a machine called a tensometer drives a plunger at constant-speed and measures the (varying) force as the plunger moves down the syringe.
The force-distance chart created in a BLEF test shows useful information: the initial break-loose force of the stationary plunger; the extrusion force (which comprises of dynamic plunger-syringe friction, and the drug formulation’s back pressure); and a small dip in force indicative of an air bubble expelled after drug delivery.
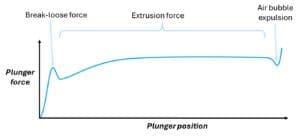
ISO 11040-8 mandates that such tests use the complete, final system ‘as intended for use’ and that the designer considers how forces vary with ageing and environmental factors. BLEF testing is valuable for characterising relationships between forces and plunger speed, syringe or needle dimensions, and fluid properties. However, it cannot be used to accurately predict injection times in an injection device because it does not reflect dynamic injection behaviour. Therefore, we need a tool that can use common BLEF test data to provide accurate predictions for injection device performance.
To address this, Sanner Group’s Design Centre of Excellence (Springboard) has co-developed advanced modelling software with Pfizer’s Devices Centre of Excellence. It allows device developers to easily forecast injection times, anticipate variation due to ageing, and set meaningful specifications early in the development process. The software can bridge the gap between constant-speed BLEF data and dynamic injection device performance without the need for guesswork, reducing the number of slow and costly prototype testing cycles and rework.
A new model and software for device performance
Software users simply upload their own BLEF data into the software and in a few steps can generate:
- A predicted distribution of injection times for their specified device,
- Maximum extrusion force requirement predictions, and
- A report for their Design History File.
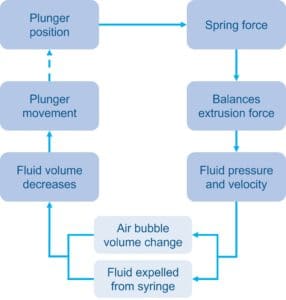
The model behind the software uses a time-stepped approach to break down an injection into discrete, calculable steps. Each loop begins with a known plunger position, which relates to the device’s spring length. From this the spring force is known and is assumed approximately equal to the current extrusion force.
Using relationships from the user’s BLEF data for the modelled syringe, the extrusion force is used to work back to the plunger speed and internal syringe pressure. By calculating the Hagen-Poiseuille equation across the needle, the pressure is related to the velocity of fluid ejected.
The plunger movement can then be calculated due to the reduction in injection volume and considering the change in any air bubble volume due to pressure changes – this is a step which is important to capture but complex and challenging to do through other predictive methods. This loop is repeated until all the volume has been delivered, and the total time elapsed gives the device injection time.
This software uses the Monte Carlo method to sample the random variation of all input parameters which include:
- User-specified tolerances on input parameters and
- The variance of real BLEF data.
Coping with device ageing
Various factors lead to device performance degradation over time, most notably desiliconisation of the syringe which leads to increased plunger-syringe friction. Device performance after shelf-life is more likely to fail requirements such as injection time than on the day the device was manufactured. This is bad news for development projects because issues might not be discovered until years into a program.
Therefore, we used newly-siliconised and desiliconised syringes as best and worst cases for plunger-syringe friction and used desiliconisation as a surrogate for ageing. We can say that syringes effectively move away from newly-siliconised performance as they age. Depending on their ageing properties, most will never get near to fully desiliconised.
We have extended the software described above to establish ageing trends using a small number of single-speed BLEF tests on aged syringes. This effect is modelled by increasing theoretical break loose and extrusion forces until devices would stall or breach injection time limits. This gives the worst-case syringes that can be allowed. Then, by back-calculating from end-of-shelf-life performance to initial conditions, the method can define a maximum allowable BLEF result at time zero. This allows engineers to specify the maximum forces allowed on a BLEF test at time zero and still be confident that the injection device would perform after shelf life.
Summary
If injection time performance is not properly predicted (including predictions for after shelf-life), a device program risks substantial cost and time overruns. The cost and time come from having to implement time-consuming and expensive component modifications and reverification testing.
Instead, we can use the new model described in this article to change the physical build-and-test loop into a mathematical model, which specifies acceptable force limits for easy BLEF tests early in the program. These predictive tools will become increasingly essential for efficient and reliable device development as drug delivery systems evolve to accommodate more demanding formulations. Our modelling software offers a practical solution by translating BLEF test data into more accurate forecasts of injection performance over a device’s lifespan.
Authors
Sanner Group:
- Matthew Latham, Consultant Engineer
- Elena Guss Tarazona, Consultant Engineer
- Bradley Sawyer, Consultant Engineer
Pfizer:
- Jay Sayed, Principal Scientist
- Charlie Bowen, Design Engineer
- Harriet Field, Laboratory Scientist Apprentice