Taking a low cost/high volume trauma device from a 3D printed prototype to an injection moulded product
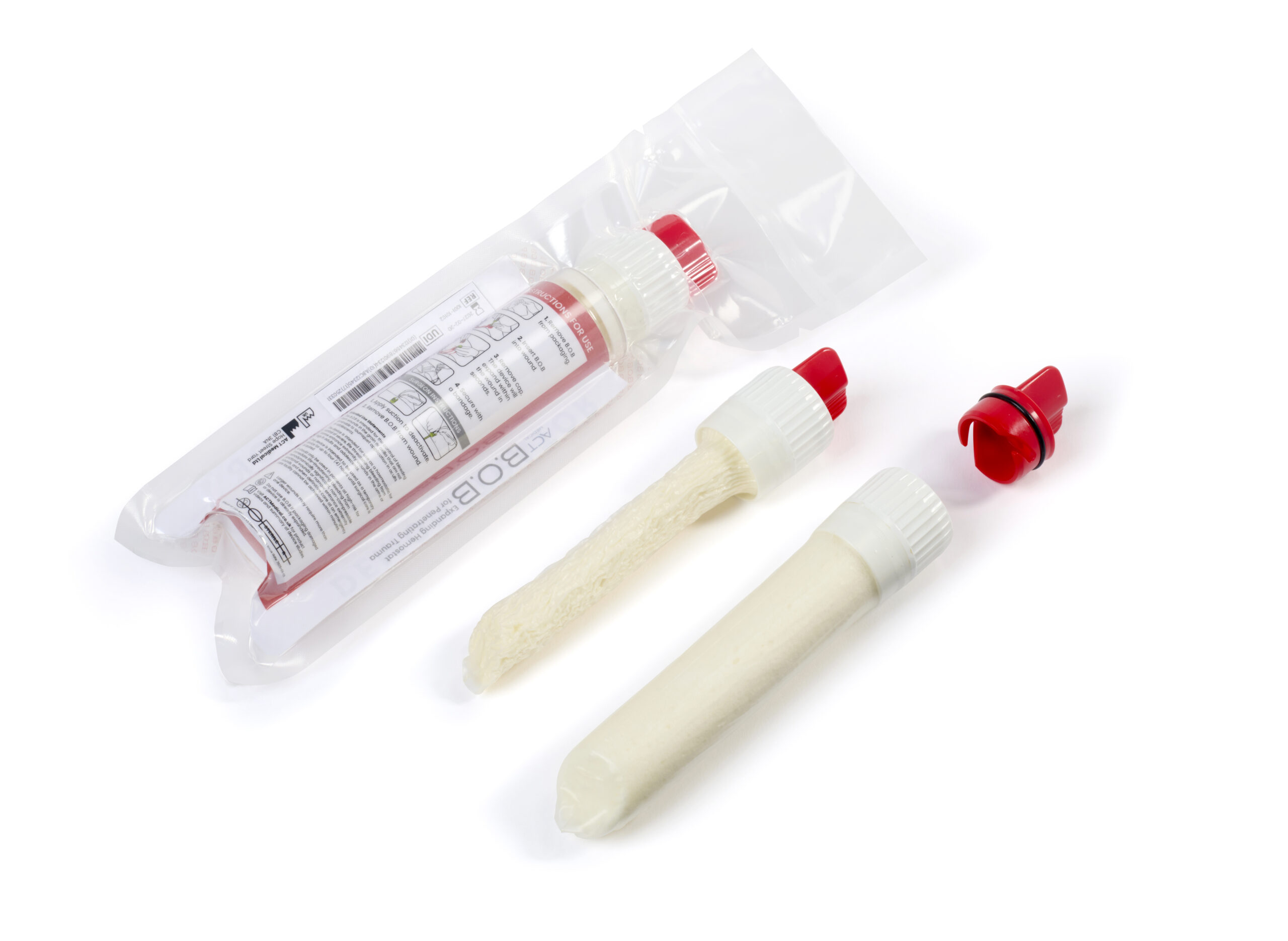
Our client, ACT Medical, are developing a novel device that aims to help first responders stem the bleeding from deep trauma wounds, such as knife or bullet wounds. When ACT Medical initially came to the Sanner Group, they had a prototype device utilising their innovative haemorrhage control concept made from 11 components, which included 3D printed parts and was manually assembled using small screws. The existing design would have been costly and challenging to produce at high volume whilst attempting to achieve ACT Medical’s low COGs target. They had also carried out a usability study which had identified some key areas for improvement with the user interface.
Over the 3-4 month period of our project working on the device, we developed an improved user interface design, designed replacement mouldable parts and a bespoke welding/assembly process, and produced injection moulded parts ready to be assembled into functional devices usable for pre-V&V testing. The final devices were made from only 6 parts, and with a fraction of the assembly time required to produce devices with a much higher reliability than the initial prototypes.
Deep penetrating trauma wounds—particularly those that result in significant haemorrhage—pose an ongoing challenge in emergency and battlefield medicine. These injuries are especially difficult to manage when they occur in anatomically complex areas such as junctional zones (e.g., the groin, armpit, or neck). In these non-compressible regions, standard haemorrhage control methods like tourniquets are often ineffective or completely unusable.
Existing haemostatic products for treating these types of wounds often suffer from limitations. Many exhibit variable efficacy depending on the wound geometry, patient condition, or application technique. Others can be cumbersome to apply quickly in high-pressure situations. A significant concern shared across many of these products is the difficulty associated with removal during surgical intervention—some materials can adhere strongly to tissue or fragment during extraction, increasing operative time and risk to the patient.
Springboard have been supporting ACT Medical in their development of a novel solution designed to overcome these limitations. Their product utilises direct internal pressure which aims to rapidly and effectively control haemorrhage at the point of injury, particularly at internal junctional bleeding sites, offering a potentially life-saving alternative where other options fall short.

Figure 1: How the device works
The development
Throughout the project, there were a few key areas which were focused on:
- The User Interface – In emergency settings, usability is paramount. We refined the product’s user interface with human factors at the forefront of our approach, through iterative ideation and in-house testing to ensure it can be deployed intuitively and consistently by first responders under stress and in challenging conditions.

Figure 2: In-house usability testing in our observation room
- Designing for Manufacture and Assembly (DFMA) at low cost/high volume – As the device is single-use and with a high volume potential market, it was key to optimise the product’s design not just for clinical efficacy, but also for scalable and reliable production. Throughout the development, a focus was kept on suitability for injection moulding and other scalable processes such as film welding, which was guided by the combined manufacturing experience of the Sanner Group.
- Producing Prototypes Using Representative Methods – Before taking the step to high volume manufacture, we utilised prototype injection moulding and worked with a supplier for film welding who was willing to work with us to produce initial low volumes but also capable of scaling up to high volume with the same design. We also developed a bespoke film-substrate welding process in-house, which allowed the devices to be assembled quickly and repeatedly and could also be scaled up to high volume. Overall, this approach to the device manufacture allowed the client to perform meaningful pre-verification and validation (pre-V&V) testing under conditions that closely mimic real-world use.
The outcome of this development phase is a more refined, production-ready product design that balances usability and effectiveness with manufacturing feasibility. These prototypes will provide the evidence needed to support investment decisions, regulatory planning, and design refinement—before committing to scaled manufacturing, potentially in partnership with a specialist manufacturer like the Sanner Group.

Figure 3: The improved design, ready for pre-V&V testing
For R&D leaders, this is a prime example of how early integration of usability testing, engineering, and manufacturing strategy can accelerate product maturity while reducing downstream risk. By making informed design decisions at this stage, our client is positioning themselves to deliver a product that is not only clinically effective but also viable at scale.